2-Weekly versus 3-weekly docetaxel to treat castration-resistant advanced prostate cancer: a randomised, phase 3 trial. | Semantic Scholar

Cancers | Free Full-Text | Effect of Baseline Characteristics on Cabazitaxel Treatment Duration in Patients with Metastatic Castration-Resistant Prostate Cancer: A Post Hoc Analysis of the Compassionate Use/Expanded Access Programs and CAPRISTANA

Pharmacokinetics, dynamics and toxicity of docetaxel: Why the Japanese dose differs from the Western dose - Kenmotsu - 2015 - Cancer Science - Wiley Online Library

2-Weekly versus 3-weekly docetaxel to treat castration-resistant advanced prostate cancer: a randomised, phase 3 trial. | Semantic Scholar
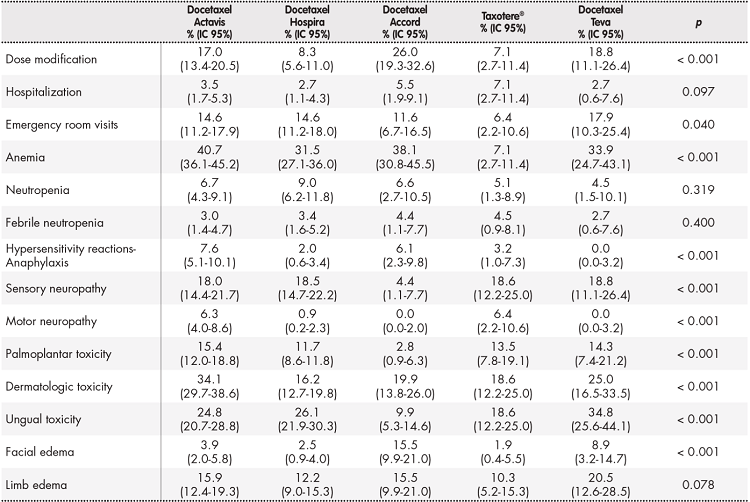
Observational study of clinical toxicity with different formulations of docetaxel in breast cancer patients